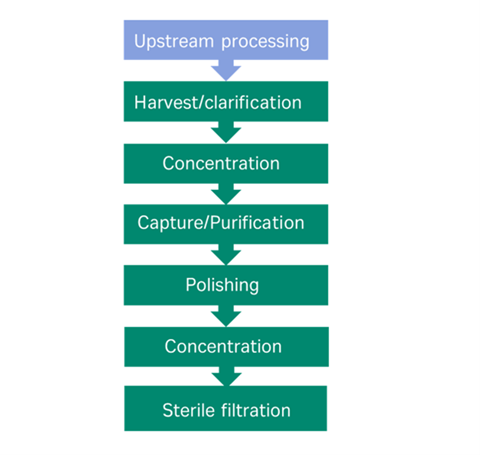
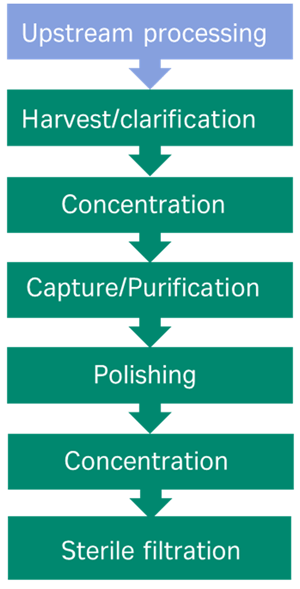
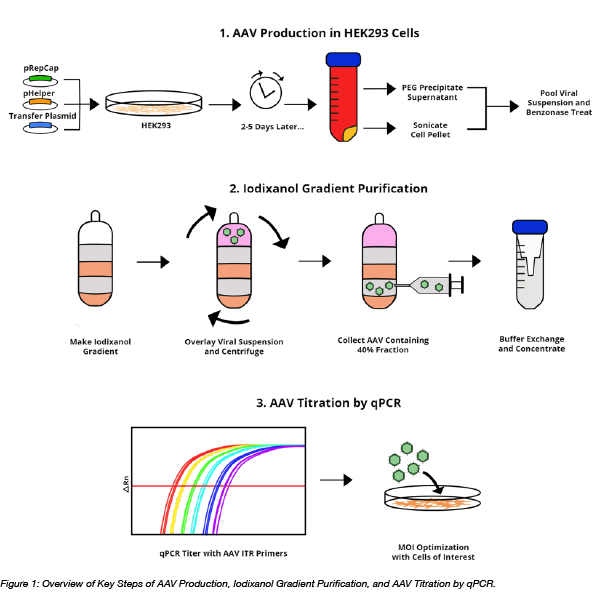
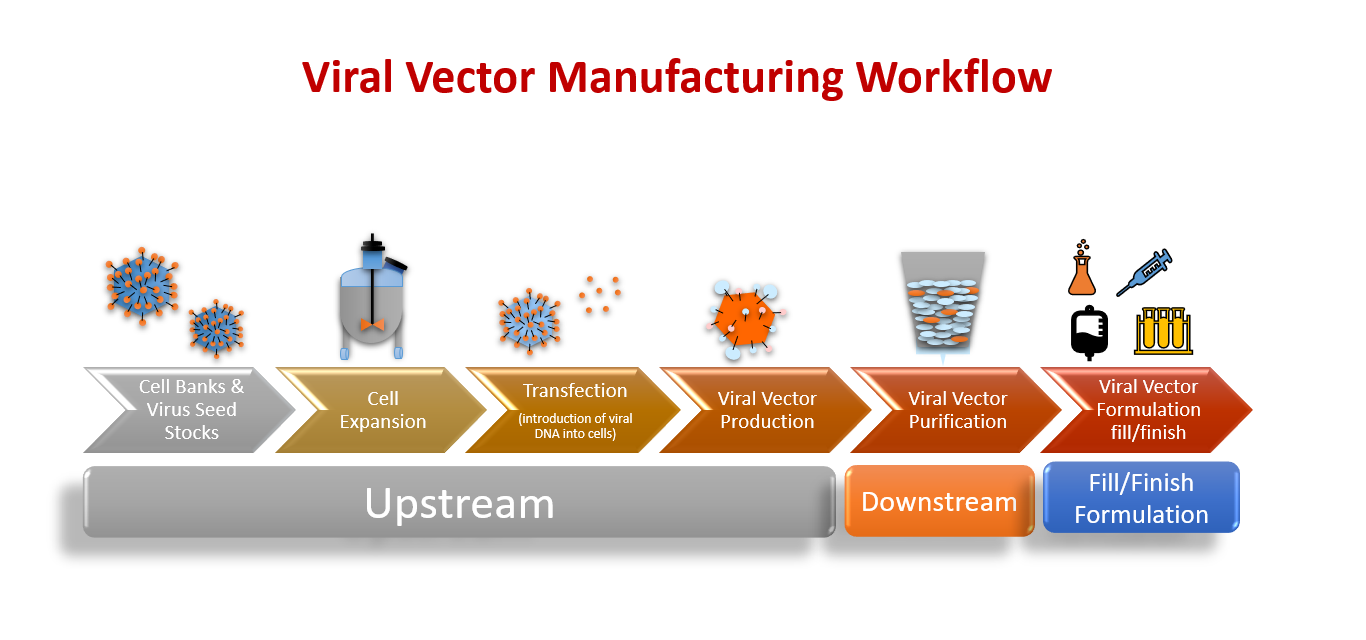
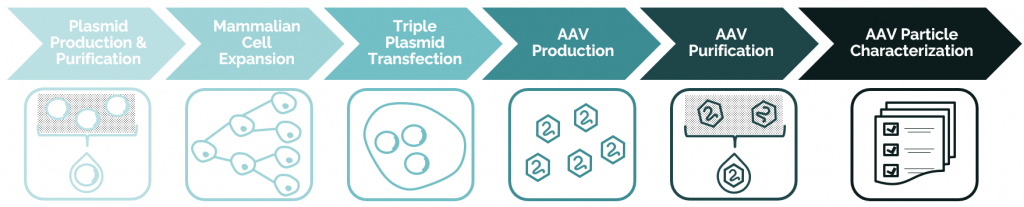
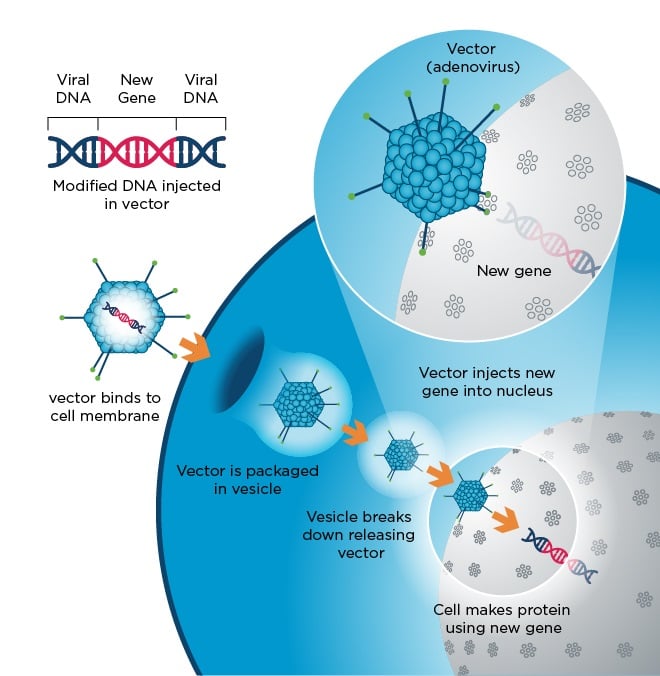
Enhancing titres of therapeutic viral vectors using the transgene repression in vector production (TRiP) system | Nature Communications

Efficient clinical-grade γ-retroviral vector purification by high-speed centrifugation for CAR T cell manufacturing: Molecular Therapy Methods & Clinical Development

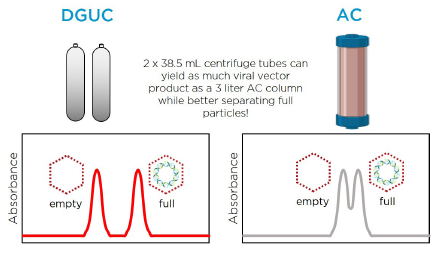
Innovative Downstream Purification Solutions for Viral Vectors: Enabling Platform Approaches to Advance Gene Therapies - BioProcess InternationalBioProcess International
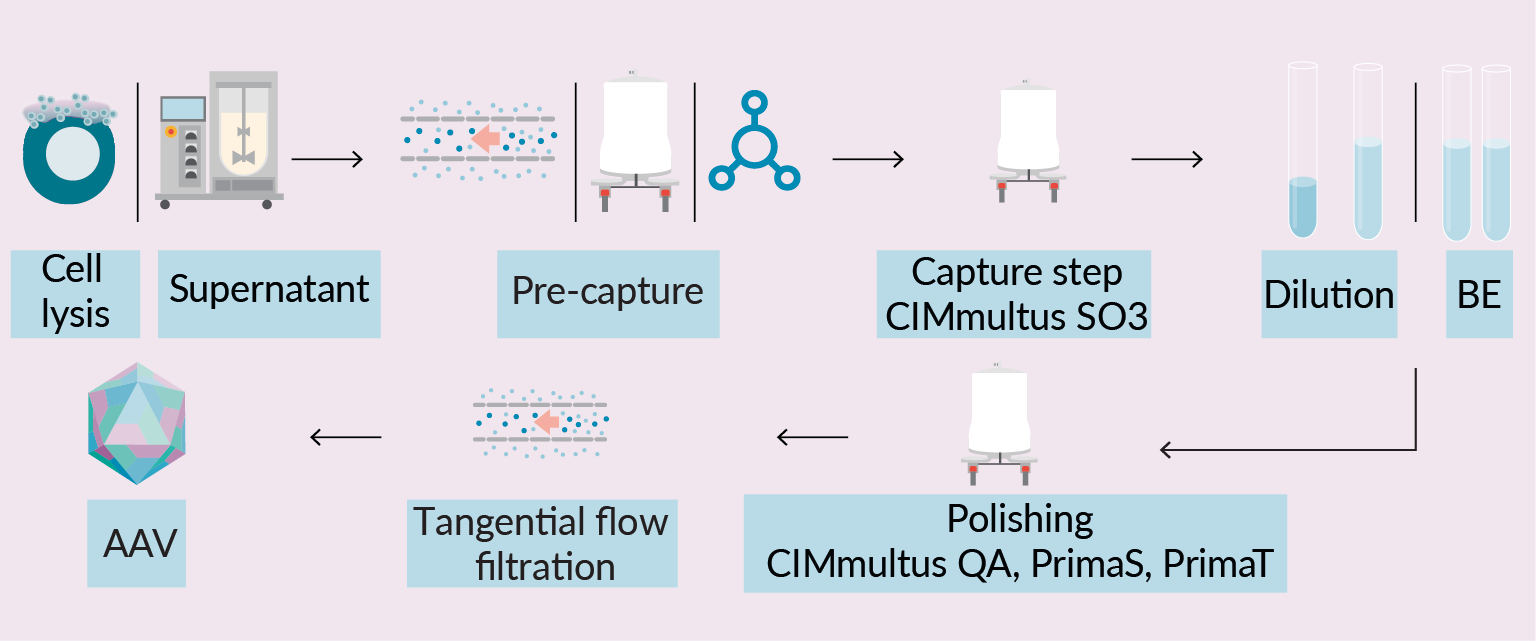
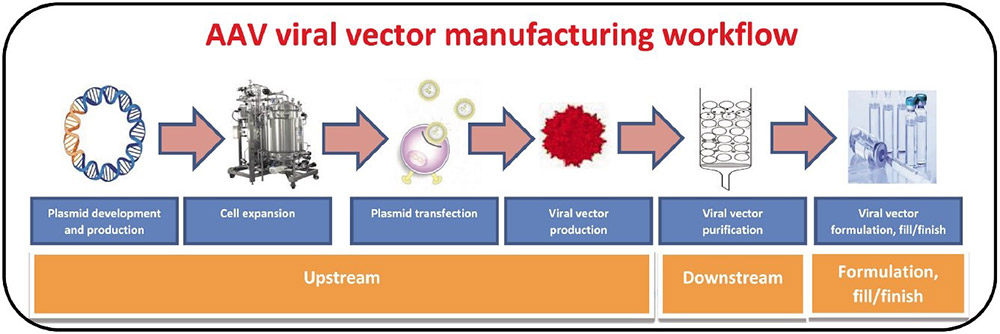
Roadmap to success in AAV purification. In-process control, high throughput & novel column modalities as necessary means for control over scalable AAV process

Laboratory-Scale Lentiviral Vector Production and Purification for Enhanced Ex Vivo and In Vivo Genetic Engineering - ScienceDirect
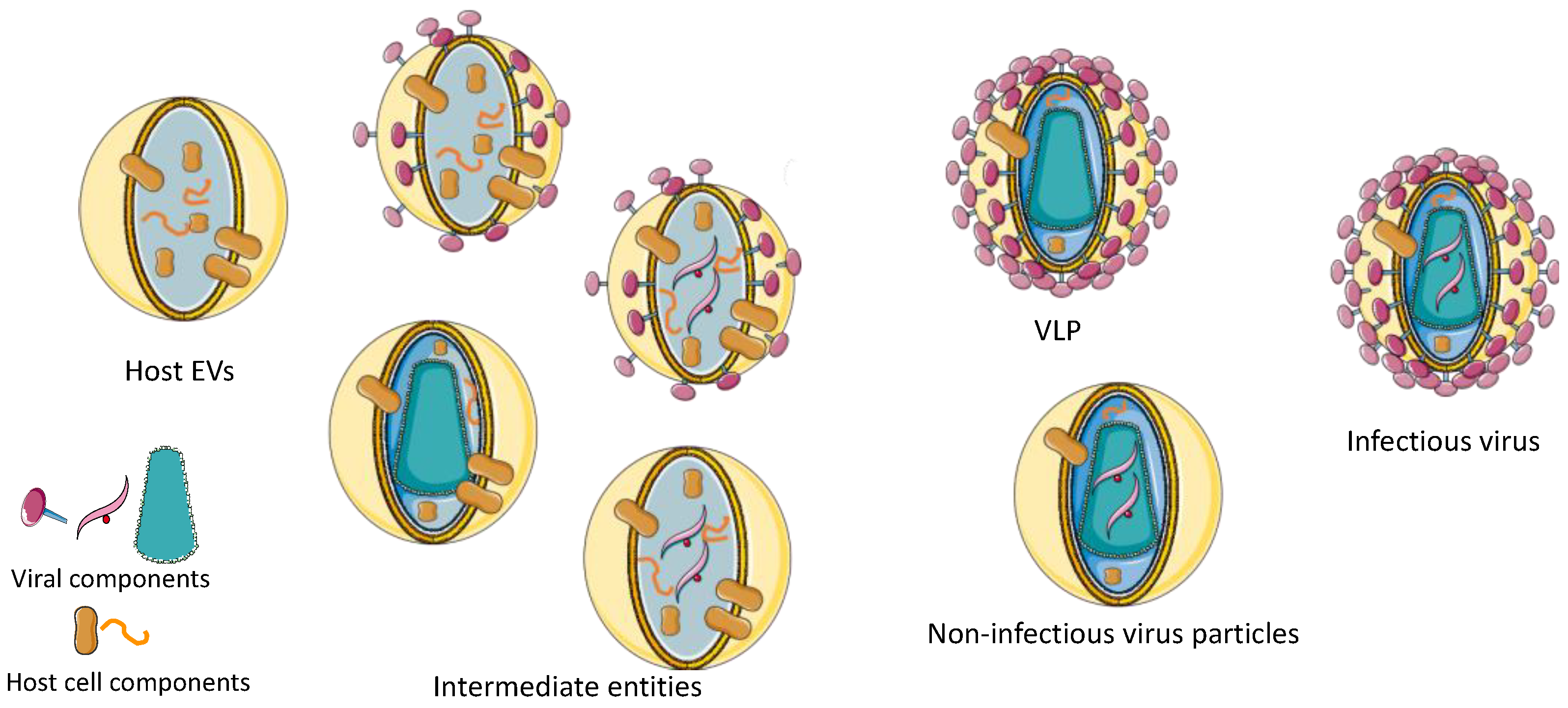
Vaccines | Free Full-Text | Critical Assessment of Purification and Analytical Technologies for Enveloped Viral Vector and Vaccine Processing and Their Current Limitations in Resolving Co-Expressed Extracellular Vesicles

Scalable Viral Vector Purification for Gene Therapy: Appraisal of Downstream Processing ApproachesBioProcess International

Figure 1 from Manufacturing of viral vectors: part II. Downstream processing and safety aspects | Semantic Scholar

Overcoming Downstream Purification Challenges for Viral Vector Manufacturing | Whitepaper | Technology Networks